Decoding HMGB1: From Molecular Mechanisms to Key "Signal Molecules" for Disease Intervention
In the microscopic world of biomedical research, a family of proteins known as "damage-associated molecular patterns" (DAMPs) is gradually emerging as a bridge connecting basic research and clinical translation. Among them, high mobility group box 1 protein (HMGB1) has attracted significant attention in the field of life sciences in recent years due to its complex functional activities, extensive signal regulatory networks, and its crucial role in various diseases. From the "guardian of genes" within the cell nucleus to the "amplifier of danger signals" outside the cell, the multiple identities of HMGB1 provide important targets for the elucidation of disease mechanisms and the development of novel therapeutic strategies.
Functional activity: The "dual role" from nuclear homeostasis to extracellular signals
The functional activity of HMGB1 exhibits a significant "position dependence", and its biological effects within cells and outside cells are completely different. This characteristic makes it a crucial switch for regulating cellular physiological and pathological processes.
Under normal physiological conditions, HMGB1 is mainly located in the cell nucleus. As a non-histone chromosomal binding protein, it participates in core physiological processes such as maintaining chromatin structure, DNA replication, transcriptional regulation, and damage repair. Studies have shown that HMGB1 can promote the binding of transcription factors to the promoter regions of target genes by bending DNA strands, thereby regulating key life activities such as cell proliferation and differentiation. It is an important "molecular chaperone" for maintaining the homeostasis of the intracellular environment.
When cells are infected, damaged or subjected to stress stimuli, HMGB1 will enter the extracellular environment through active secretion or passive release. At this point, its function rapidly switches to that of a "danger signal molecule". The HMGB1 that enters the extracellular space will bind to the receptors on the surface of immune cells through its unique domains, activating the immune response and inflammatory reaction, and initiating the body's damage repair mechanism. It is worth noting that the activity of extracellular HMGB1 is also strictly regulated by post-translational modifications. Modifications such as acetylation, phosphorylation and methylation can affect its nuclear export efficiency and receptor binding ability, further enriching the complexity of its functional regulation.
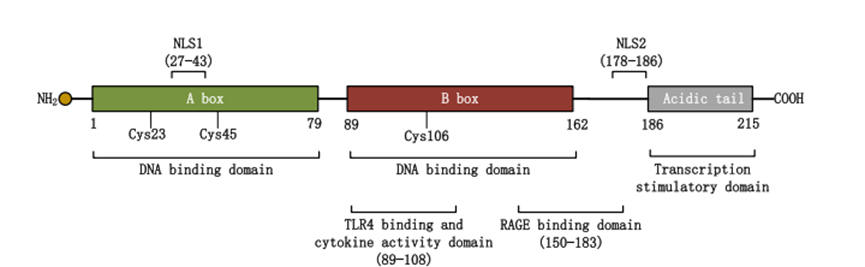
Signal pathway: The "core regulatory network" linking immunity and inflammation
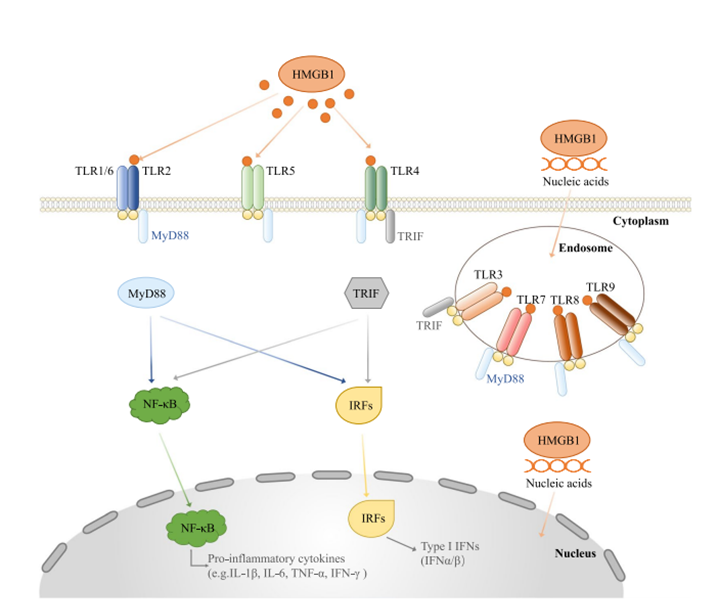
The extracellular biological effects of HMGB1 are mainly achieved by activating multiple signaling pathways. These pathways interweave to form a complex regulatory network, jointly participating in pathological physiological processes such as immune response, inflammatory reaction, and tissue damage. Among them, the binding of HMGB1 to advanced glycation end products receptor (RAGE) and Toll-like receptors (TLRs) is the core mechanism by which HMGB1 initiates signal transduction.
The binding of HMGB1 to RAGE can activate downstream mitogen-activated protein kinase (MAPK) and nuclear factor-κB (NF-κB) signaling pathways. The activation of the MAPK pathway can promote the transcription and release of inflammatory factors, while NF-κB, as a classic inflammatory regulator, can further amplify the inflammatory signal, recruit immune cells to the injury site, and accelerate the progression of the inflammatory response. In the tumor microenvironment, the continuous activation of this pathway also promotes tumor angiogenesis and the invasion and metastasis of tumor cells.
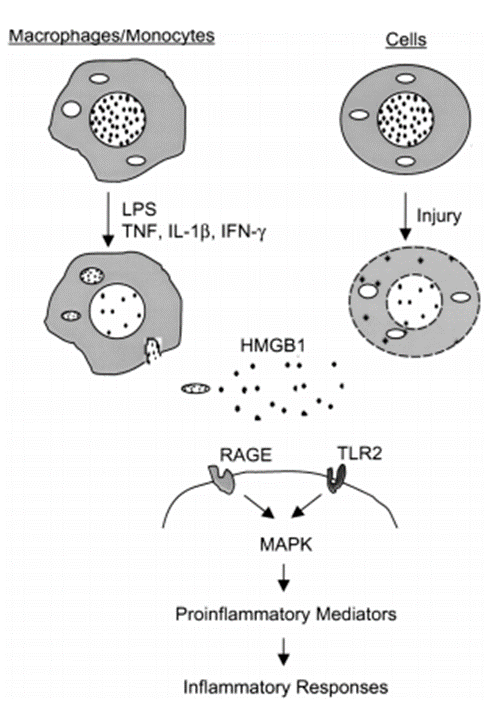
In addition, HMGB1 can also bind to receptors such as TLR2 and TLR4, and activate the innate immune response via the MyD88-dependent signaling pathway. This process not only exerts a crucial anti-infective effect in infectious diseases, but also disrupts immune tolerance in autoimmune diseases, leading to damage of autologous tissues. Studies have demonstrated that interventions targeting the HMGB1 signaling pathway, such as the use of RAGE antagonists or TLR inhibitors, can effectively suppress the excessive activation of inflammatory responses, providing novel insights for the treatment of related diseases.
Disease Effects: The "Multifaceted Regulatory Factor" from Inflammation to Tumors
Due to its complex functional activity and signal regulation network, HMGB1 plays a crucial role in the occurrence and development of various diseases, covering areas such as inflammatory diseases, tumors, and neurological disorders, and has become a highly promising diagnostic marker and therapeutic target in clinical research.
In acute inflammation and infectious diseases, the release level of HMGB1 is closely related to the severity of the disease. In patients with sepsis, HMGB1, as a late inflammatory factor, the increase in its serum concentration often indicates a poor prognosis. Animal experiments have shown that neutralizing the activity of HMGB1 can significantly reduce the mortality rate of sepsis model mice, providing a new direction for the treatment of sepsis. In chronic inflammatory diseases such as rheumatoid arthritis, HMGB1 continuously activates inflammatory signaling pathways, promoting the proliferation of synovial cells and the release of inflammatory factors, aggravating the destruction of joint tissues. Targeted treatment against HMGB1 has shown good anti-inflammatory effects in preclinical studies.
In the field of oncology, the role of HMGB1 exhibits a "dual nature". On one hand, in the early stage of tumor occurrence, the release of HMGB1 can activate anti-tumor immune responses and inhibit tumor growth; on the other hand, as the tumor progresses, tumor cells will actively secrete HMGB1, which, by activating the RAGE-NF-κB pathway, promotes tumor angiogenesis, the formation of an immunosuppressive microenvironment, and epithelial-mesenchymal transition (EMT) of tumor cells, accelerating tumor invasion and metastasis. Currently, the detection of HMGB1 expression levels in tumor tissues or serum has been used for the prognosis assessment of some tumors, and the combined application of HMGB1 inhibitors and immunotherapy has also become a research hotspot in the field of tumor treatment.
In neurological disorders, the abnormal release of HMGB1 is closely related to the pathological processes of diseases such as ischemic stroke and Alzheimer's disease. After ischemic stroke occurs, the HMGB1 released by damaged nerve cells will activate the inflammatory response, aggravating the ischemia-reperfusion injury of brain tissue; while in the brain tissue of Alzheimer's disease patients, the interaction between HMGB1 and β-amyloid protein will promote the formation of senile plaques and accelerate the progression of neurodegenerative diseases.
Conclusion: Targeting HMGB1, opening up a new dimension in disease treatment
From the basic physiological functions within the cell nucleus to the pathological signal regulation outside the cell, HMGB1's multiple roles make it an indispensable and important molecule in biomedical research. With the continuous deepening of research on its functional activity, signaling pathways, and disease mechanism, the value of HMGB1 in disease diagnosis and treatment is gradually becoming more prominent. Whether as a "barometer" of disease progression or as a "breakthrough point" for targeted therapy, HMGB1 provides broad space for clinical translational research. It is believed that in future research, with the further decoding of the regulatory mechanism of HMGB1, new diagnostic technologies and treatment plans centered on HMGB1 will continue to emerge, bringing new hope to more patients with diseases.
Functional activity: The "dual role" from nuclear homeostasis to extracellular signals
The functional activity of HMGB1 exhibits a significant "position dependence", and its biological effects within cells and outside cells are completely different. This characteristic makes it a crucial switch for regulating cellular physiological and pathological processes.
Under normal physiological conditions, HMGB1 is mainly located in the cell nucleus. As a non-histone chromosomal binding protein, it participates in core physiological processes such as maintaining chromatin structure, DNA replication, transcriptional regulation, and damage repair. Studies have shown that HMGB1 can promote the binding of transcription factors to the promoter regions of target genes by bending DNA strands, thereby regulating key life activities such as cell proliferation and differentiation. It is an important "molecular chaperone" for maintaining the homeostasis of the intracellular environment.
When cells are infected, damaged or subjected to stress stimuli, HMGB1 will enter the extracellular environment through active secretion or passive release. At this point, its function rapidly switches to that of a "danger signal molecule". The HMGB1 that enters the extracellular space will bind to the receptors on the surface of immune cells through its unique domains, activating the immune response and inflammatory reaction, and initiating the body's damage repair mechanism. It is worth noting that the activity of extracellular HMGB1 is also strictly regulated by post-translational modifications. Modifications such as acetylation, phosphorylation and methylation can affect its nuclear export efficiency and receptor binding ability, further enriching the complexity of its functional regulation.

Signal pathway: The "core regulatory network" linking immunity and inflammation
The extracellular biological effects of HMGB1 are mainly achieved by activating multiple signaling pathways. These pathways interweave to form a complex regulatory network, jointly participating in pathological physiological processes such as immune response, inflammatory reaction, and tissue damage. Among them, the binding of HMGB1 to advanced glycation end products receptor (RAGE) and Toll-like receptors (TLRs) is the core mechanism by which HMGB1 initiates signal transduction.
The binding of HMGB1 to RAGE can activate downstream mitogen-activated protein kinase (MAPK) and nuclear factor-κB (NF-κB) signaling pathways. The activation of the MAPK pathway can promote the transcription and release of inflammatory factors, while NF-κB, as a classic inflammatory regulator, can further amplify the inflammatory signal, recruit immune cells to the injury site, and accelerate the progression of the inflammatory response. In the tumor microenvironment, the continuous activation of this pathway also promotes tumor angiogenesis and the invasion and metastasis of tumor cells.

In addition, HMGB1 can also bind to receptors such as TLR2 and TLR4, and activate the innate immune response via the MyD88-dependent signaling pathway. This process not only exerts a crucial anti-infective effect in infectious diseases, but also disrupts immune tolerance in autoimmune diseases, leading to damage of autologous tissues. Studies have demonstrated that interventions targeting the HMGB1 signaling pathway, such as the use of RAGE antagonists or TLR inhibitors, can effectively suppress the excessive activation of inflammatory responses, providing novel insights for the treatment of related diseases.

Disease Effects: The "Multifaceted Regulatory Factor" from Inflammation to Tumors
Due to its complex functional activity and signal regulation network, HMGB1 plays a crucial role in the occurrence and development of various diseases, covering areas such as inflammatory diseases, tumors, and neurological disorders, and has become a highly promising diagnostic marker and therapeutic target in clinical research.
In acute inflammation and infectious diseases, the release level of HMGB1 is closely related to the severity of the disease. In patients with sepsis, HMGB1, as a late inflammatory factor, the increase in its serum concentration often indicates a poor prognosis. Animal experiments have shown that neutralizing the activity of HMGB1 can significantly reduce the mortality rate of sepsis model mice, providing a new direction for the treatment of sepsis. In chronic inflammatory diseases such as rheumatoid arthritis, HMGB1 continuously activates inflammatory signaling pathways, promoting the proliferation of synovial cells and the release of inflammatory factors, aggravating the destruction of joint tissues. Targeted treatment against HMGB1 has shown good anti-inflammatory effects in preclinical studies.
In the field of oncology, the role of HMGB1 exhibits a "dual nature". On one hand, in the early stage of tumor occurrence, the release of HMGB1 can activate anti-tumor immune responses and inhibit tumor growth; on the other hand, as the tumor progresses, tumor cells will actively secrete HMGB1, which, by activating the RAGE-NF-κB pathway, promotes tumor angiogenesis, the formation of an immunosuppressive microenvironment, and epithelial-mesenchymal transition (EMT) of tumor cells, accelerating tumor invasion and metastasis. Currently, the detection of HMGB1 expression levels in tumor tissues or serum has been used for the prognosis assessment of some tumors, and the combined application of HMGB1 inhibitors and immunotherapy has also become a research hotspot in the field of tumor treatment.
In neurological disorders, the abnormal release of HMGB1 is closely related to the pathological processes of diseases such as ischemic stroke and Alzheimer's disease. After ischemic stroke occurs, the HMGB1 released by damaged nerve cells will activate the inflammatory response, aggravating the ischemia-reperfusion injury of brain tissue; while in the brain tissue of Alzheimer's disease patients, the interaction between HMGB1 and β-amyloid protein will promote the formation of senile plaques and accelerate the progression of neurodegenerative diseases.
Conclusion: Targeting HMGB1, opening up a new dimension in disease treatment
From the basic physiological functions within the cell nucleus to the pathological signal regulation outside the cell, HMGB1's multiple roles make it an indispensable and important molecule in biomedical research. With the continuous deepening of research on its functional activity, signaling pathways, and disease mechanism, the value of HMGB1 in disease diagnosis and treatment is gradually becoming more prominent. Whether as a "barometer" of disease progression or as a "breakthrough point" for targeted therapy, HMGB1 provides broad space for clinical translational research. It is believed that in future research, with the further decoding of the regulatory mechanism of HMGB1, new diagnostic technologies and treatment plans centered on HMGB1 will continue to emerge, bringing new hope to more patients with diseases.











